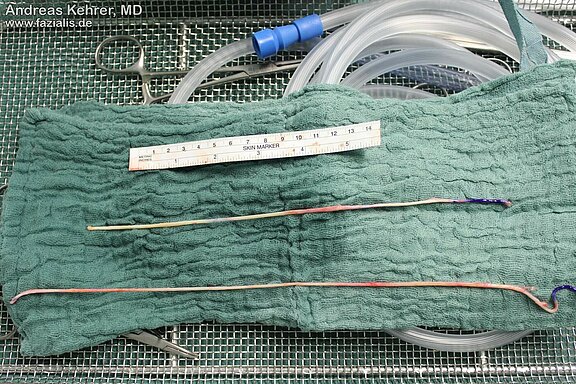
One procedure we frequently perform to reanimate a paralyzed half of the face is the transfer of nerves using 'cross-face-nerve-grafts' (CFNGs). In order to bridge the distance, a suitable nerve graft is required, mostly skin nerves from the calf, which are removed without any significant loss of sensation via very small additional incisions on the lower leg.
The most elegant surgical option for reconstructing facial nerve palsy in the early phase is neurotization. The most common procedure is known as neuro-neural neurorotization. Here a functionally dispensable healthy nerve branch is connected with the functionless nerve branch of a muscle or muscle group to be reinnervated. This makes it possible to reconstruct the original motor function, i.e. to reactivate a paralyzed muscle in the early phase of its paralysis. Figuratively speaking, "new electricity is supplied to disconnected but intact electric motors (muscles) via an alternative power cable (new nerve connection)".
In the early stage of facial paralysis (about 6-15 months after onset), nerve transfers and possibly additional nerve grafts for neurotization are used. Even if the facial nerve fails, the existing, functionally intact musculature can at best be completely preserved and protected from transformation into functionless fatty tissue (atrophy) if new nerve impulses are supplied to it. This is possible even if the mimetic musculature does not receive any impulses at all from the original facial nerve, e.g. if its main trunk had to be removed with a tumor removal (acoustic neuroma).
Donor nerve branches from the facial nerve system from the non-paralyzed half of the face are often used. This requires an extension of the corresponding donor nerve by a nerve graft ("extension cable") from the healthy to the paralyzed facial side; this is referred to as the creation of a "cross-face nerve graft" (CFNG). The advantage of this surgical method is that it uses the functioning facial nerves of the healthy side without reducing its function. The facial nerve of the non-paralyzed half of the face has enough fibers to deliver some nerve branches that are strong enough to also control the diseased side. This is done without without causing visible functional deficits in the healthy half of the face through their "misuse". CFNGs enable spontaneity and an emotional coupling of the function to be restored by impulses from the other half of the face. As the name suggests, the two nervous systems are "bridged" by the nerve graft. Nerve grafts are usually taken from superficial skin nerves via small inconspicuous additional incisions on the lower leg. This means that minimal loss of sensation must be expected locally.
If the mimetic musculature, which is still viable but paralyzed without nerve stimulus, can be "reactivated" in this early interval of facial paresis, the best results can be expected with regard to important facial functions such as eye closure, facial expression, control of the corner of the mouth, and near complete restoration of facial symmetry. This is called "facial reanimation". The amount of newly introduced nerve fibers ("current strength") is a critical factor in determining the percentage of the original performance of the mimetic muscles ("electric motors") that can be restored.
In order to reanimate the paralyzed half of the face, additional methods can be selected or used in combination. Donor nerves from the paralyzed half of the face are also used. Their function is usually not impaired in peripheral facial paresis, in central forms (e.g. after stroke) their function must be tested beforehand. On the one hand, fibres of the nerve to the masseter muscle (Nervus massetericus) of the same half of the face can be repositioned and control the mimetic musculature. In a very similar procedure, other nerves in the paralyzed facial hemisphere with physiologically different functions can also be used for this purpose (e.g. hypoglossal nerve, "tongue nerve").
Figure 1
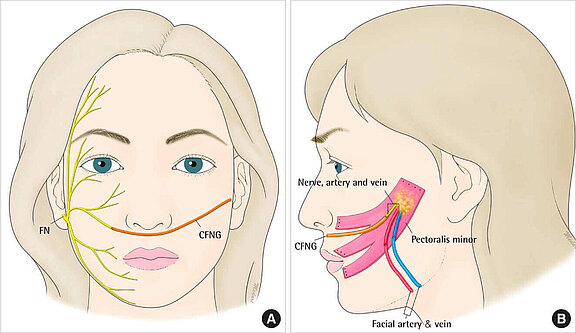
The figure shows the sural nerve (calf skin nerve) as a CFNG to bridge the distance from the healthy right half of the face to the diseased left side. The graft redirects selected nerve fibers of the intact facial nerve into the paralyzed half of the face. The right picture shows a free muscle graft of the pectoral minor muscle (musculus pectoralis) and its innervation by the CFNG.
Source: Leckenby J, Grobbelaar A. Archives of Plastic Surgery 2013;40(5):633-638. Epub on 09/13/2013. DOI: doi.org/10.5999/aps.2013.40.5.633. CC BY-NC 3.0. creativecommons.org/licenses/by-nc/3.0/.
In this surgical procedure, the "masticatory muscle nerve" or one of its lower branches (masseteric nerve) is used to reinnervate the mimetic musculature. The masticatory muscle nerve branch is located in the paralyzed half of the face and originates from another nerve, the fifth cranial nerve, called the trigeminal nerve. It is not affected by a loss of function in peripheral facial nerve palsy. However, in the case of central facial paresis (e.g. after a stroke), it can also fail.
The masticatory muscle nerve is particularly suitable for use as a donor nerve for neurotizing procedures. It is also used in selected indications as a "impulse generator" for freely transplanted functional muscles. The masseteric nerve, as the lower branch of the masticatory muscle nerve system, carries a particularly large number of nerve fibers (axons), which is why it can provide a large proportion of these without a functional deficit.
A 'V-to-VII transfer' is an operative procedure in which the masseteric nerve, the fifth cranial nerve (Roman V.), is connected to the functionally impaired facial nerve (seventh cranial nerve, Roman VII.). Its nerve fibers orient themselves after the rearrangement now at the functionless facial nerve as a guide rail and reach over it to reach the mimetic musculature. As soon as the masticatory muscle nerve fibers have reached the target musculature, it is neurotized, i.e. supplied with new nerve impulses. If "current" now flows in the nerve branch system of the masticatory muscle nerve, the mimetic muscles also receive "current", take up their function, and are reactivated. In most cases, the patient has to bite a little in order to initiate the newly gained function. Through learning processes that can be supported by physiotherapy, occupational therapy, and/or speech therapy exercises, many patients can integrate the new function into their facial expressions in such a way that biting is no longer necessary over time. Over time, the brain "notices" that the branched nerve fibers have now been given a new task (e.g. smiling) and assigns this new function to them in "superordinate control centers". This cognitive rewiring is referred to as "cerebral plasticity".
This 22-year-old young patient suffered from a complete facial paralysis on the left side after tumor surgery (no continuity of the facial nerve). On the left picture a distinct flaccid orbicularis oculi muscle is visible, the palpebral fissure is distinctly wide despite the attempt to smile. The cheek shows no muscle tension (tonus), speech is slightly slurred. The corner of the mouth on the left hangs and no smile line (nasolabial fold) can be formed. After nerve transfer (V-to-VII) and transplantation of multiple nerve grafts (CFNG) in one operation, the symptoms are already improved a few months later (5-6 months): there is clearly more tone of the orbicularis oculi (eyelid fissure already narrower), the patient can again arbitrarily control the corner of the mouth to smile and the nasolabial fold is visible. His speech is clear again. Neurotization of the original mimic muscles by the nerve reconstruction will still progress and symmetry, facial expression and function will further improve.
Thus, the learning ability (plasticity) of the brain contributes to a natural smile if this operative method is chosen.[1],[2] Many patients can consciously distinguish between the activation of the masticatory muscle and the activation of the mimetic musculature after this operation procedure.[3]
Disadvantages of this method can be synkinesis (unintentional mass movements) during the chewing process, e.g. a slight smile or winking of the eyes when chewing during food intake. The movements are often only very subtly visible, so that in practice there are no problems worth mentioning in everyday life and the patients cope well with them. Spontaneity and emotional coupling are not as reliable with V-to-VII transfer as with CFNG, where the facial nerve of the healthy half of the face functions as the donor nerve.
Murphy et. al were able to show in a study that V-to-VII transfer is associated with low morbidity. This study also showed that an average time of 5.76 months is required after a V-to-VII transfer until reinnervation occurs and dynamics and resting tone of the face are restored.[4]
Reinnervation by cross-face nerve transplants (CFNG) is slower, but is functionally much more specific with appropriate selection of donor nerve branches. Since "current", i.e. nerve fiber capacity, is lost on the long distance between both halves of the face, we very often combine both procedures. Thus, we carry out V-to-VII nerve transfer as well as nerve grafting with two CFNGs in combination. In this way, the "best of two donor nerve regions" can be meaningfully combined in terms of selectivity of the desired functions and muscle strength through high axon capacity ("current strength").
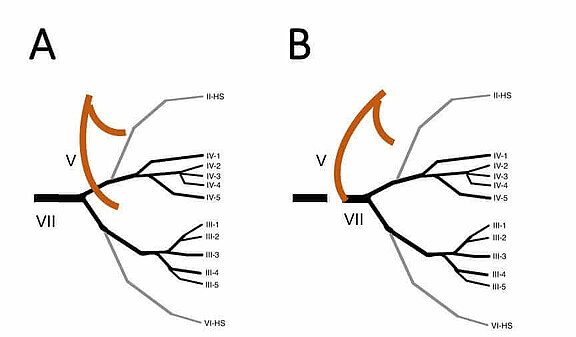
Figure A:
The massteric nerve (V) presents itself with a horizontal and a descending branch. The facial nerve (VII) divides into several branches, including a temporozygomatic main branch and a cervicofacial main branch, before dividing into further sub-branches. Anatomically seen, the nerves run in different tissue layers, but still have an anatomical proximity and can therefore be easily coapted.
Figure B:
In the V-to-VII transfer, the descending branch of the masseteric nerve (V) is coapted with the main trunk of the facial nerve (VII). In addition to this procedure, a cross-face nerve-graft procedure can be used if required [5].
The transfer of the masticatory muscle nerve to the facial nerve-controlled musculature is a very efficient procedure in "facial reanimation" (restoration of facial expressions). Usually, patients of advanced age are not granted a high potential of nerve regeneration by nerve transfers. In this sprightly 84-year-old patient, a renewed static/dynamic suspension procedure on the temporal muscle (externally pre-operated patient) was combined with an additional masticatory nerve transfer. The function of the suspension procedure is immediately detectable postoperatively and showed, as expected, an improved symmetry of rest and only a moderate excursion of the corner of the mouth. At a further presentation at six to nine months later, the "smile musculature" suddenly appears to be controllable by the masticatory muscle nerve rearrangement (V to VII transfer). The masticatory muscle nerve has "delivered" its branched nerve fibres to the still intact cheek muscles and now spontaneously controls a smile. The positively surprised patient was prescribed ergotherapy and speech therapy to further improve his mimic coordination.
sources:
[1] Klebuc MJ. Facial reanimation using the masseter-to-facial nerve transfer. Plast Reconstr Surg. 2011;127(5):1909-15.
[2] Manktelow RT, Tomat LR, Zuker RM, Chang M. Smile reconstruction in adults with free muscle transfer innervated by the masseter motor nerve: effectiveness and cerebral adaptation. Plast Reconstr Surg. 2006;118(4):885-99.
[3] Hontanilla B, Marre D. Differences between sexes in dissociation and spontaneity of smile in facial paralysis reanimation with the masseteric nerve. Head Neck. 2014;36(8):1176-80.
[4] Murphey AW, Clinkscales WB, Oyer SL. Masseteric Nerve Transfer for Facial Nerve Paralysis: A Systematic Review and Meta-analysis. JAMA Facial Plast Surg. 2018;20(2):104-10.
[5] Kehrer A, Engelmann S, Ruewe M, Geis S, Taeger C, Kehrer M, Tamm ER, Bleys RLAW, Prantl L, Mandlik V. Perfusion maintains functional potential of denervated mimic muscles in early persistent facial paralysis which requires early microsurgical treatment - the histoanatomic basis of the extratemporal facial nerve trunk assessing axonal load in the context of possible nerve transfers. Clin Hemorheol Microcirc. 2018;70(1):1-13. doi: 10.3233/CH-189905.